PHARMACOLOGY
COURSE 010
Testosterone Drug Formulations: Injections, Transdermal Agents, Intranasal Gel, Pellets, and Oral Formulations
Learn testosterone drug formulation including injections, transdermals, pellets, and oral agents, and why matching the right delivery system to the patient's lifestyle, adherence patterns, and pharmacokinetic needs is one of the most consequential decisions in testosterone therapy.
PROGRAM Advanced Clinical Training Program, Testosteronology Society™
ESTIMATED TIME TO COMPLETE 45 Minutes Reading + 30 Minute Video
TARGET AUDIENCE Clinicians treating patients with testosterone deficiency, androgen-related conditions, and broader hormonal health challenges.
COURSE FORMAT Evidence-informed clinical education module including text, video, and diagrams
FACULTY DISCLOSURE Authors and reviewers include Thomas O’Connor, M.D., and Omar Hajmousa, PharmD. This educational material is designed as an independent clinical training course. Formal CME accreditation can not currently be claimed.
CONTENT DISCLOSURE This educational material was developed with editorial assistance from AI technology and then reviewed, revised, and verified by the Testosteronology Society™ faculty to ensure accuracy, clinical appropriateness, and educational value.
COURSE PREREQUISITES Courses are structured to be taken in sequence so clinicians can build a coherent clinical framework. Individual courses may be revisited anytime for refresher learning or when a patient presentation relates to a topic addressed in the curriculum.
IMPORTANT NOTE The course material is provided for general educational and informational purposes only. It is not intended to serve as medical advice, diagnosis, or treatment recommendations for any specific individual.
Course Overview
Drug formulation choice is one of the most important clinical decisions in testosterone therapy beyond just laboratory values. The same weekly dose can feel very different depending on the pharmacokinetic profile of the chosen formulation and patient adherence to the regimen. The cost of the drug itself is also an important variable. Current guidelines and literature support the idea that route, timing, adherence, and monitoring all materially influence both symptom stability and safe follow-up. [1-4]
This course reviews testosterone formulations through a practical clinical lens. The emphasis is on pharmacokinetic reasoning, adherence realities, lab timing, and side-effect interpretation. A central theme is that many apparent testosterone problems can be formulation problems, especially when volatility, poor technique, missed doses, or inconsistent monitoring are mistaken for treatment failure. [1-5]
Learning Objectives
After completing this course, clinicians should be able to:
➀ Explain how pharmacokinetics, peaks, troughs, and steady state influence testosterone therapy outcomes.
➁ Compare common testosterone drug formulation using practicality, tolerability, and monitoring needs.
➂ Recognize when symptoms reflect drug formulation instability rather than inadequate total exposure.
➃ Match a testosterone formulation to patient lifestyle, adherence patterns, and clinical risk.
➄ Apply a structured approach to monitoring and troubleshooting patient problems regarding the formulations.
Course Topics
The following topics will be covered in the course text, video, diagrams or downloadable documents:
➀ Why Drug Formulation Choice Drives Outcomes
➁ Core Pharmacokinetics, Lab Timing, And Steady State
➂ Injectable Testosterone and Pellets: Esters, Intervals, And Route
➃ Transdermal Therapies: Gels, Creams, And Patches
➄ Oral Testosterone Formulations
➅ Matching Formulation To Patient Lifestyle And Adherence
➆ Monitoring, Side Effects, And Switching Strategies
“The delivery system is not a minor detail after the diagnosis is made. It is one of the main determinants of whether treatment will feel smooth, chaotic, convenient, unsustainable, or unsafe.”

Thomas O'Connor, M.D.
010 Testosterone Drug Formulations: Injections, Transdermal Agents, Pellets, and Oral Formulations
27 MINUTE COURSE TRAINING VIDEO
With Thomas O'Connor, M.D. Founder / CEO Testosteronology Society™
Video Lesson Takeaways
TO BE INCLUDED AFTER VIDEO IS RECORDED
COURSE TEXT
➀ Why Drug Formulation Choice Drives Outcomes
In clinical practice, formulation choice often determines whether testosterone therapy feels stable or disruptive. Patients usually do not present by saying their average exposure is inadequate. They describe fatigue late in the dosing interval, irritability after administration, sleep disruption, edema, acne flares, or a sense that treatment works briefly and then fades. Those complaints are often clues to the shape of the exposure curve rather than proof that the weekly dose is too low. Clinical guidance and formulation reviews consistently emphasize that route and schedule affect symptom pattern, adherence, and interpretability of follow-up. [1-4]
A useful mindset is to treat delivery-system selection as a clinical matching problem. A formulation that fits a patient’s schedule, tolerance, privacy needs, and follow-up capacity is more likely to produce stable care than one that looks ideal on paper but repeatedly fails in real life. Patients with chaotic routines may struggle with daily transdermal therapy. Patients sensitive to peak-trough shifts may do poorly with longer injection intervals. Patients who can’t adhere to the prescribed regimen can become quietly inconsistent, which can then trigger unnecessary dose escalation. [1-4]
➁ Core Pharmacokinetics, Lab Timing, And Steady State
Pharmacokinetics of the chosen formulation and timing of labs matter because testosterone levels change over time after administration, and those changes can shape both symptoms and lab interpretation. High peaks may cause adverse effects and make testosterone levels look supratherapeutic on paper, while low troughs may give the patient a feeling that therapy “wears off” before the next dose. Mapping symptom occurrence relative to the dosing regimen can help provide clinical context and evidence to decision making. Combining symptom patterns with laboratory values completes the clinical picture. As a general guideline, draw and interpret labs at the nadir (trough) and use that as the basis for dose adjustments. [5]
Reaching steady-state is crucial for decision making. If route, dose, and frequency are adjusted too quickly, clinicians end up reading noise rather than signal. For that reason, dose and formulation changes should usually be interpreted only after a reasonable stabilization window, with the timing of follow-up labs and symptom review clearly documented. Generally, steady-state is reached after 5 half-lives. For example, testosterone cypionate has a half-life of 8 days and day 40 or 6 weeks would be considered steady-state. This is especially important when clinicians want the chart to remain intelligible for future providers. [1-4]
Below is a graph of drug X concentration versus time with an assumption of a 7 day half-life. The drug is dosed once weekly. Notice that steady-state is where the drug concentration waves consistently between a certain peak and trough.
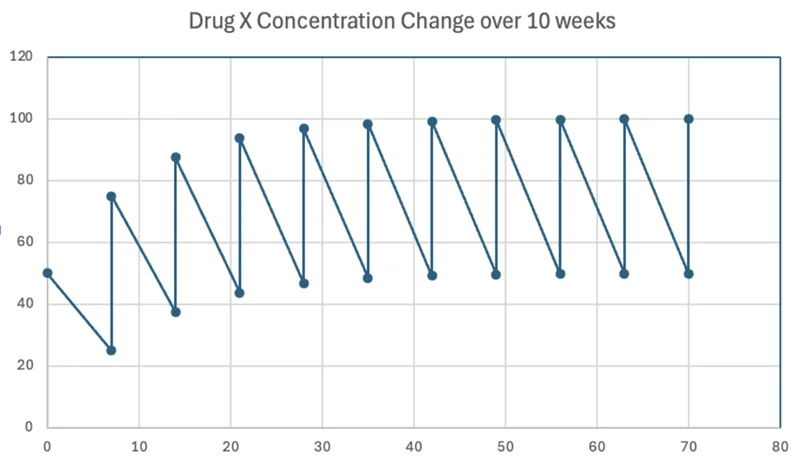
Figure 1. Conceptual representation of drug accumulation and steady-state pharmacokinetics assuming a 7-day half-life.
➂ Injectable Testosterone and Pellets: Esters, Intervals, And Route
Injectable testosterone cypionate and enanthate remain some of the most widely used treatment options because of their relatively known pharmacokinetic profile, cost, and availability. While their long half-life might be a strength, it can also be a weakness due to the potential for high peaks and low troughs if dosed improperly at any interval above 1 week. One mistake clinicians can make is assuming the patient knows how to measure, administer, and store their medication. Educating the patient on how to properly complete those steps is crucial to ensuring a regimen that is effective for the patient. [5]
Subcutaneous administration can be more comfortable and easier to administer for some patients and evidence points towards comparable pharmacokinetics to intramuscular administration. While there is no evidence on subcutaneous administration being superior in efficacy, it can be another tool in your arsenal to use if the patient is intimidated by intramuscular administration. [6]
Injectable testosterone undecanoate provides a long-acting testosterone formulation designed for extended dosing intervals under supervised office administration due to the black box warning for pulmonary oil microembolism and anaphylaxis. Its pharmacokinetic profile allows maintenance of therapeutic testosterone concentrations with dosing approximately every 10 weeks after the initial loading phase. While the long duration of action can improve adherence and reduce injection frequency, it also limits dose flexibility and rapid adjustment if adverse effects occur. Having to administer in office and cost can also limit the ability to utilize this formulation. [7]
Pellets appeal to patients who want fewer administration events and less weekly or daily treatment friction. Their main advantage is adherence support through infrequent dosing. Their main limitation is low adjustability once implanted. Other risks include procedural issues such as extrusion, infection, bruising, and local discomfort, which is why pellets fit best in patients who value convenience and accept slower response to any needed adjustment. [5]
➃ Transdermal Therapies
Transdermal gels and creams can work very well when absorption and daily routine are consistent. Their major strength is smoother day-to-day exposure compared with wider-interval regimens and their utilization in patients who have a fear of needles. Their major weakness is variability and cost. Skin conditions, sweating, bathing, application habits, and simple inconsistency can all change delivery enough to create unstable symptom narratives. Transfer to female household members and children is another risk. Transdermal failure is often behavioral or situational before it is pharmacologic. Before assuming the dose is inadequate, clinicians should assess adherence to the regimen. This kind of counseling is not minor. It is often the difference between a formulation that looks ineffective and one that works predictably. [5]
Patches reduce transference concerns compared with gels, but they bring their own burden. Adhesion problems, visibility, and local skin irritation are common reasons patients stop using them consistently. If the patch does not stay on well, or the skin becomes irritated, the patient is effectively receiving an erratic regimen even when the prescription is correct. [5]
➄ Oral Testosterone Formulations
Oral testosterone has resurfaced as a therapy option recently. Older oral testosterone formulations were historically limited by hepatotoxicity due to 17-ɑ-alkylation. Newer oral testosterone undecanoate products use different delivery strategies through the lymphatic system and now have FDA-approved roles in appropriate patients. Some limitations of oral testosterone undecanoate include food timing, dosing frequency, and cost. [10-13]
*See supplemental materials for details on dosing, frequency, and clinical pearls for these formulations.
Reference guide to available testosterone delivery systems, dosing, and clinical pearls.
| Generic | Brand | Strength | Route | Half Life | Dosing | Pearls |
|---|---|---|---|---|---|---|
| Injectable | ||||||
| Testosterone Cypionate1*Compounding available with various carrier oils and excipients | Depo-Testosterone Azmiro |
200 mg/mL Or 100 mg/mL (less common) |
IM/SubQ | ~8 days | Starting dose for most men 70–100 mg every 5–7 days. Dose on an individual basis but frequency should be every 7 days at the longest. |
|
| Testosterone Enanthate2*Compounding available with various carrier oils and excipients | Delatestryl Xyosted |
200 mg/mL | IM/SubQ | 4.5–7 days IM* References unclear |
Dose on an individual basis but frequency should be every 7 days at the longest. Xyosted: Start 75 mg SubQ every 7 days. Target nadir 350–650 ng/dL total T. Increase by 25 mg/week if <350 and decrease by 25 mg/week if trough ≥650 ng/dL. |
|
| Testosterone Propionate14*Compounding available with various carrier oils and excipients | Commercially not available in the USA | 100 mg/mL | IM | 2–3 days per dosing regimen | 25–50 mg 2–3 times every week or 50–100 mg every other day in male athletes. | All information from Llewellyn's Anabolics 11th Edition — not a typical medical text. |
| Testosterone Undecanoate3 | Aveed Nebido (abroad) |
250 mg/mL | IM | ~3 months | 750 mg ×1 then 750 mg after 4 weeks (loading), then 750 mg every 10 weeks.5 In Europe/Abroad: 1 gm IM Q3 months. |
|
| Testosterone Esters5 | Sustanon 250 *Not available in the USA |
250 mg/mL | IM | Variable due to variety of esters | Pkg insert: Every 3 weeks Should be injected every 7 days14 |
|
| Pellets | ||||||
| Testosterone4 | Testopel | 75 mg/pellet | SubQ | Variable | 150–450 mg every 3–6 months6 |
|
| Topical / Transdermal | ||||||
| Testosterone Gel6*Compounding Available. Scrotal gel also available through compounding — note disproportionately elevated DHT levels due to high 5-alpha-reductase activity in the scrotum.17 | Androgel / Vogelxo / Testim | 1% Androgel: 25 mg/2.5 g or 50 mg/5 g Vogelxo: 50 mg/5 g or 12.5 mg/pump Testim: 50 mg/5 g |
Topical | Steady state by day 2–3 | 50 mg QAM to shoulders and upper arms or abdomen; increase to 75 mg or 100 mg based on response. |
|
| Testosterone Pump6*Compounding Available | Androgel 1.62% | 20.25 mg/pump 20.25 mg/1.25 g or 40.5 mg/2.5 g |
Topical | — | 40.5 mg QAM to shoulders and upper arms or abdomen. Dose adjustments in 20.25 mg increments w/ the pump. |
|
| Testosterone Gel7*Compounding Available | Fortesta | 2% 10 mg/0.5 g |
Topical | — | 40 mg QAM to the thighs. Dose adjustments in 10 mg increments w/ the pump. |
|
| Testosterone Topical Solution8*Compounding Available | Axiron | 30 mg/1.5 mL | Topical | Steady state within 14 days | 60 mg QAM to the axilla using the applicator. Dose adjustments in 30 mg increments w/ the pump. |
|
| Testosterone Transdermal Solution9*Historically available as a scrotal patch. No longer on the market. | Androderm | 2 mg/day or 4 mg/day | Transdermal | 70 minutes | 4 mg Androderm system applied to the back, upper arms, abdomen or thighs QPM. Dose adjustments in 2 mg/day increments using the 2 mg patches. |
|
| Intranasal | ||||||
| Testosterone Nasal Gel10 | Natesto | 5.5 mg/0.122 g | Intranasal | — | 2 actuations (11 mg) TID |
|
| Oral | ||||||
| Testosterone Undecanoate Oral Capsule11,12,13 | Jatenzo / Kyzatrex / Tlando | Jatenzo: 158, 198, 237 mg Kyzatrex: 100, 150, 200 mg Tlando: 112.5 mg |
PO | 2 hours | Jatenzo: 158–396 mg BID with food Kyzatrex: 100–400 mg BID with food Tlando: 225 mg BID with food |
|
Or 100 mg/mL (less common)
Starting dose for most men 70–100 mg every 5–7 days. Frequency should be every 7 days at the longest.
- Gold Standard for TRT. Cheap and easy to access.
- Potential for high peaks and low troughs due to long half-life.
- Microdosing over a shorter interval can help with adverse effects but increases injection burden.
- Compounded variations can help with excipient and dose optimization.
References unclear
Frequency should be every 7 days at the longest. Xyosted: Start 75 mg SubQ every 7 days. Target nadir 350–650 ng/dL total T. Adjust ±25 mg/week based on trough.
- Insurance issues may arise with Xyosted if prescribed more frequently than every 7 days.
- Xyosted auto-injector is convenient but can be challenging to fine-tune dose.
25–50 mg 2–3 times/week or 50–100 mg every other day in male athletes.
All information from Llewellyn's Anabolics 11th Edition — not a typical medical text.
750 mg ×1, then 750 mg at 4 weeks (loading), then 750 mg every 10 weeks.5 In Europe: 1 gm IM Q3 months.
- Administered in office by a healthcare professional.
- Patients should be observed for 30 mins after administration.
Pkg insert: Every 3 weeks. Should be injected every 7 days.14
- Formulated in peanut oil — check for ALLERGIES.
- Contains 30 mg/mL propionate, 60 mg/mL phenyl propionate, 60 mg/mL isocaproate, 100 mg/mL decanoate.
150–450 mg every 3–6 months.6
- Pellets may extrude from the skin.
- Very difficult to adjust dose.
- 2 pellets for each 25 mg testosterone propionate required weekly.4
50 mg QAM to shoulders, upper arms, or abdomen. Increase to 75–100 mg based on response.
- Good option for fear of needles.
- Allow area to dry before dressing. Flammable.
- Avoid swimming/showering for at least 5 hours.
- Avoid contact with women or children.
40.5 mg QAM to shoulders, upper arms, or abdomen. Adjustments in 20.25 mg increments w/ the pump.
- Allow area to dry before dressing. Flammable.
- Avoid swimming/showering for at least 5 hours.
- Avoid contact with women or children.
40 mg QAM to the thighs. Adjustments in 10 mg increments w/ the pump.
- Allow area to dry before dressing. Flammable.
- Avoid swimming/showering for at least 2 hours.
- Avoid contact with women or children.
60 mg QAM to the axilla using the applicator. Adjustments in 30 mg increments w/ the pump.
- Flammable. Avoid swimming/showering for at least 2 hours.
- Apply deodorant BEFORE applying Axiron.
4 mg system applied to back, upper arms, abdomen, or thighs QPM. Adjustments in 2 mg/day increments using the 2 mg patches.
- Avoid swimming/showering for at least 2 hours.
- Itching is the most common adverse event.
- Fold patch in half to discard in household trash.
2 actuations (11 mg) TID
- TID dosing with Cmax of 1,044 ng/dL (40 mins post-dose) and Cmin of 215 ng/dL.
- Weak evidence of preserving fertility.15,16
- Discontinue temporarily with severe rhinitis.
Jatenzo: 158–396 mg BID with food
Kyzatrex: 100–400 mg BID with food
Tlando: 225 mg BID with food
- Different formulations are not interchangeable.
- No hepatotoxicity noted.
- PO is the easiest route of administration for adherence.
- See package inserts for recommended dosing and titrations.
- Testosterone cypionate injection [package insert]. Northstar Rx/AccessData. December 2022.
- XYOSTED (testosterone enanthate) [package insert]. AccessData (FDA). 2019.
- AVEED (testosterone undecanoate) [prescribing information]. AveedUSA.com. Latest.
- Testopel (testosterone pellets) [package insert]. Endo USA, Inc. Latest.
- Sustanon 250 (testosterone mixture) [patient information, UK]. Medicines.org.uk / EMC. Latest.
- AndroGel (testosterone gel) [package insert]. AccessData (FDA). 2013.
- Fortesta (testosterone topical gel) [dosage guide]. Drugs.com. ~July 2025.
- Axiron (testosterone topical solution) [product monograph]. AccessData (FDA) & DailyMed. Latest.
- Androderm (testosterone transdermal system) [prescribing information]. AccessData (FDA). May 2020.
- Natesto (testosterone nasal gel) [full prescribing information]. Natesto.com. ~2019.
- Jatenzo (oral testosterone undecanoate) [prescribing information]. Jatenzo.com. ~2023.
- Kyzatrex (oral testosterone undecanoate) [prescribing information]. AccessData (FDA). ~2022.
- Tlando (oral testosterone undecanoate) [prescribing information]. Tlando.com. Feb 2024.
- Llewellyn W. Anabolics. 11th ed. Jupiter, FL: Molecular Nutrition; 2017.
- Naelitz BD, Momtazi-Mar L, Vallabhaneni S, et al. Testosterone replacement therapy and spermatogenesis in reproductive age men. Nat Rev Urol. 2025;22(10):703–719.
- Ramasamy R, Masterson TA, Best JC, et al. Effect of Natesto on Reproductive Hormones, Semen Parameters and Hypogonadal Symptoms. J Urol. 2020;204(3):557–563.
- Korenman SG, Viosca S, Garza D, et al. Androgen therapy of hypogonadal men with transscrotal testosterone systems. Am J Med. 1987;83(3):471–478.
➅ Matching Formulation To Patient Lifestyle And Adherence
Formulation matching should begin with the patient’s actual day-to-day life. Travel patterns, shift work, privacy, skin sensitivity, tolerance for injections, household transfer risk, cost, and ability to return for monitoring all shape whether a regimen is likely to succeed. Choosing the right formulation for the patient reduces preventable instability and makes future interpretation more defensible because the clinical reasoning is clear from the start.
➆ Monitoring, Side Effects, And Switching Strategies
Monitoring should take into account all the variables discussed above. Injection regimens need a clearly defined point in the dosing interval for repeat labs, the nadir. Transdermal regimens need consistent timing relative to application. Pellet regimens are interpreted across longer horizons. Oral regimens need timing consistency. [1-4]
Side effects should be interpreted through pattern recognition whenever possible. Edema, irritability, insomnia, acne, and perceived wearing-off symptoms may each have kinetic fingerprints. When symptoms cluster after administration, the peak may need to be softened. When symptoms cluster late in the interval, adherence or interval length may be the main issue. When complaints do not track with dosing at all, clinicians should widen the lens to sleep, stress, stimulants, psychiatric factors, underground lab androgen use, and medical comorbidities before assuming testosterone is the primary cause. [1-4]
Switching is best handled in a staged way. Define the problem clearly, change one major variable at a time, reset the monitoring rules, and allow a stable observation window before judging the result. This approach reduces attribution confusion and preserves continuity when patients later move between clinicians or clinics. It also keeps the record far more useful than a sequence of rapid, overlapping changes. [1-4]
COURSE SUMMARY
Testosterone formulation choice plays a major role in treatment outcomes because it affects pharmacokinetics, adherence, symptom stability, and lab interpretation. Injectable, transdermal, pellet, and oral therapies can all be effective, but each has distinct advantages and limitations related to dosing intervals, absorption, adjustability, and patient routine. Many apparent treatment failures are actually problems of formulation mismatch, inconsistent administration, or poorly timed monitoring rather than inadequate dosing. Clinicians who understand peak–trough dynamics, steady-state timing, and adherence challenges are better able to interpret symptoms and make rational adjustments. Matching the formulation to the patient’s lifestyle and monitoring consistently helps produce safer and more stable testosterone therapy.
REFERENCES
- Bhasin S, Brito JP, Cunningham GR, et al. Testosterone therapy in men with hypogonadism: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2018;103(5):1715-1744. doi:10.1210/jc.2018-00229
- Mulhall JP, Trost LW, Brannigan RE, et al. Evaluation and management of testosterone deficiency: AUA guideline. J Urol. 2018;200(2):423-432. doi:10.1016/j.juro.2018.03.115
- Hackett G, Kirby M, Edwards D, et al. British Society for Sexual Medicine guidelines on adult testosterone deficiency, with statements for UK practice. J Sex Med. 2017;14(12):1504-1523. doi:10.1016/j.jsxm.2017.10.067
- Boeri L, Masterson T, Antonio L, et al. Testosterone Therapy in Adult Males with Hypogonadism. Eur Urol. Published online December 24, 2025. doi:10.1016/j.eururo.2025.12.015
- Shoskes JJ, Wilson MK, Spinner ML. Pharmacology of testosterone replacement therapy preparations. Transl Androl Urol. 2016;5(6):834-843. doi:10.21037/tau.2016.07.10
- Spratt DI, Stewart II, Savage C, et al. Subcutaneous injection of testosterone is an effective and preferred alternative to intramuscular injection. J Clin Endocrinol Metab. 2017;102(7):2349-2355. doi:10.1210/jc.2017-00359
- Endo Pharmaceuticals Inc. AVEED® (testosterone undecanoate) injection, for intramuscular use: Prescribing Information. Malvern, PA: Endo Pharmaceuticals Inc; 2025.
- Swerdloff RS, Wang C, White WB, et al. A New Oral Testosterone Undecanoate Formulation Restores Testosterone to Normal Concentrations in Hypogonadal Men. J Clin Endocrinol Metab. 2020;105(8):2515-2531. doi:10.1210/clinem/dgaa238
- U.S. Food and Drug Administration. AVEED (testosterone undecanoate) injection prescribing information. Endo Pharmaceuticals Inc; 2023.
- U.S. Food and Drug Administration. JATENZO (testosterone undecanoate) capsules prescribing information. Clarus Therapeutics Inc; 2019. Updated periodically.
- U.S. Food and Drug Administration. TLANDO (testosterone undecanoate) capsules prescribing information. Antares Pharma Inc; 2022.